Preparing a buffy coat from whole blood is one of the most useful and cost-effective steps laboratories can perform when working with immune cells. Whether the goal is leukocyte isolation, PBMC enrichment, or downstream cell separation workflows, the buffy coat offers a concentrated, cleaner starting material compared to whole blood.
In many labs, buffy coat is sourced directly from blood banks as a byproduct of blood donation processing. However, there are many situations where researchers need to prepare buffy coat in-house, working with fresh samples, limited volumes, experimental blood collections, or time-sensitive workflows. In these cases, knowing how to generate a reliable buffy coat fraction becomes essential.
Buffy coat preparation does not require complex chemistry or specialized equipment. It relies on physical separation principles, mainly controlled centrifugation, to concentrate leukocytes between plasma and red blood cells. When done correctly, the result is a highly enriched leukocyte fraction that can be used directly for cell isolation, enrichment of rare populations, or functional assays.
This article provides a practical, step-by-step guide to preparing buffy coat from whole blood in your own laboratory. It explains what buffy coat is, how it forms, what materials and conditions are required, how to avoid common mistakes, and how to use the resulting fraction for downstream cell isolation workflows.
Understanding Buffy Coat and Its Role in Cell Isolation
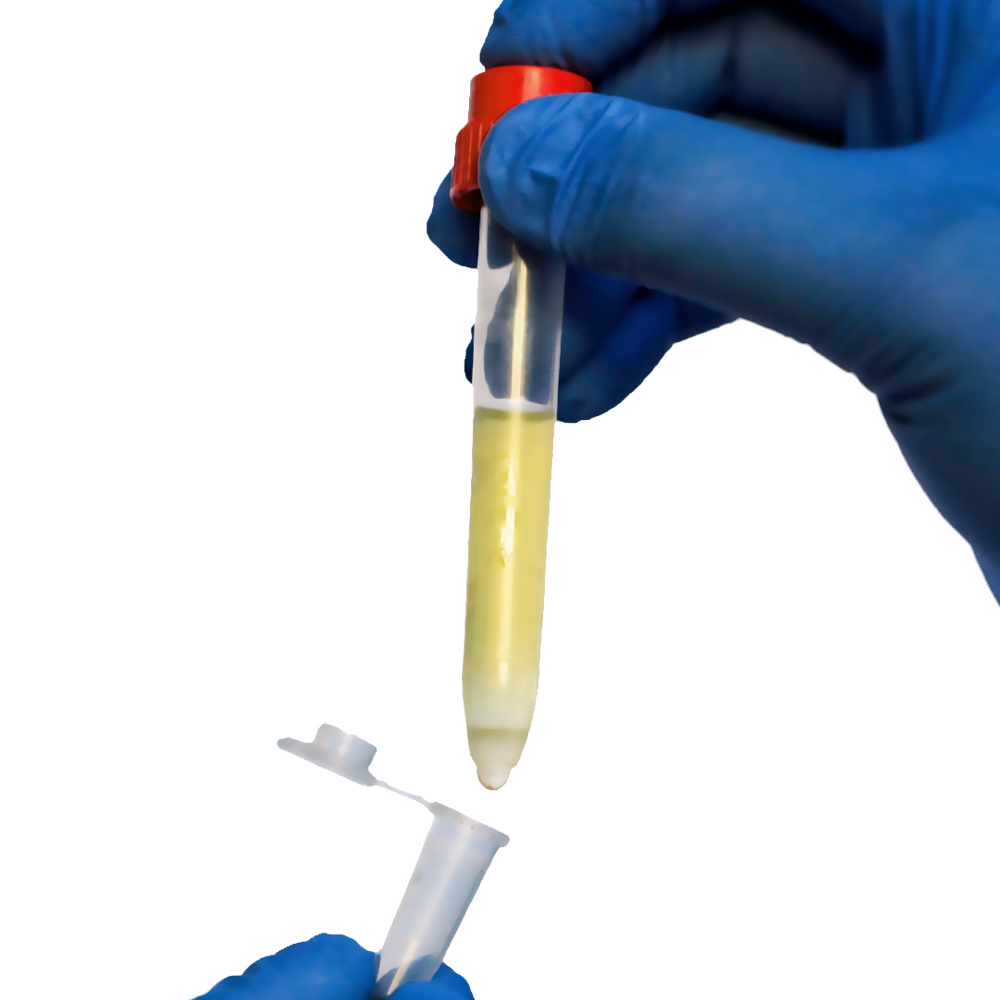
A buffy coat, also known as a leukocyte concentrate, is the thin, pale layer that forms between plasma and red blood cells after whole blood is centrifuged. This layer contains a high concentration of white blood cells, including lymphocytes, monocytes, and other immune cells, along with a smaller number of platelets.
In whole blood, leukocytes make up only a small fraction of the total volume. Preparing a buffy coat concentrates these cells by 10–20 times compared to their original concentration in blood. This makes buffy coat an ideal starting material for cell isolation workflows, especially when working with rare cell types or when high cell numbers are needed quickly.
Buffy coat plays an important role in both clinical and research contexts. In blood banks, leukocytes are removed from blood products to reduce transfusion reactions and improve storage stability. The removed leukocyte fraction, the buffy coat, is then available as a resource for research and cell isolation.
For laboratory workflows, buffy coat offers several advantages over whole blood:
- Higher leukocyte concentration
- Lower red blood cell content
- Reduced sample volume
- Faster downstream processing
Because buffy coat preparation is based on physical separation rather than chemical treatment, the cells remain viable and functional. This makes buffy coat especially suitable for applications such as immune cell enrichment, functional assays, and multi-step cell separation strategies.
Materials, Conditions, and Preparation Before You Start
Before preparing buffy coat in your lab, it is important to ensure that basic conditions and materials are in place. While the protocol itself is straightforward, consistency depends on preparation.
Whole Blood Requirements
- Use freshly collected whole blood whenever possible
- Blood should be anticoagulated (e.g., EDTA, citrate, or heparin)
- Avoid clotted or hemolyzed samples
Fresh blood generally produces cleaner layer separation. However, buffy coat can still be prepared from blood that is several hours old, provided handling is gentle and centrifugation conditions are adjusted carefully.
Buffers and Tubes
- Washing buffer (e.g., PBS or cell wash buffer)
- Standard centrifuge tubes (15 mL or 50 mL, depending on volume)
- Pipettes and sterile pipette tips
Centrifuge Setup
- Swing-bucket rotor (preferred for clear layer formation)
- Ability to control speed, time, and brake settings
- Room temperature operation
Environmental Conditions
- Perform preparation at room temperature
- Avoid temperature fluctuations during centrifugation
- Minimize delays between steps
Proper preparation ensures that the buffy coat layer forms cleanly and can be collected without contamination from plasma or red blood cells.
Step-by-Step Protocol to Prepare Buffy Coat in the Lab
Preparing buffy coat in your own lab can be done using a simple centrifugation protocol. While the steps themselves are straightforward, attention to detail at each stage is what determines yield, purity, and reproducibility. The protocol below is widely used in research laboratories and works well for fresh whole blood collected with standard anticoagulants.
Step 1: Dilute the Whole Blood
- Mix one part whole blood with one part washing buffer
- Gently invert to mix
- Avoid vigorous shaking to prevent cell damage
Diluting whole blood before centrifugation serves several important purposes. Whole blood is naturally viscous due to its high red blood cell content, and dilution reduces this viscosity, allowing cells to separate more cleanly during centrifugation. A gentler separation improves the visibility and integrity of the buffy coat layer.
Inversion should be slow and controlled. Vigorous mixing can activate platelets or stress leukocytes, which may affect downstream applications. The goal is uniform dilution without introducing bubbles or shear forces.
Step 2: Load the Centrifuge Tubes
- Transfer diluted blood into centrifuge tubes
- Balance tubes carefully
- Ensure caps are secure
Proper tube loading is critical for consistent layer formation. Uneven volumes or poorly balanced tubes can lead to uneven centrifugal force, resulting in distorted layers or partial mixing. Always use matched tubes with equal volumes and place them symmetrically in the rotor.
Ensure that tube caps are fully closed. Leakage during centrifugation not only compromises the sample but can contaminate the centrifuge and disrupt neighboring tubes.
Step 3: Centrifugation
- Centrifuge at 200 × g for 10 minutes
- Use a swing-bucket rotor
- Brake OFF to avoid disturbing layers
Swing-bucket rotors are preferred because they allow layers to form horizontally, creating a clean and easily identifiable buffy coat interface. Fixed-angle rotors can compress layers and make collection more difficult.
Turning the brake off is essential. Sudden deceleration causes turbulence that can remix plasma, buffy coat, and red blood cells. Allow the centrifuge to decelerate naturally to preserve layer integrity.
After centrifugation, three distinct layers should be visible:
- Top: Plasma
- Middle: Buffy coat (thin, white layer)
- Bottom: Red blood cells
Step 4: Identify the Buffy Coat Layer
The buffy coat appears as a cloudy or whitish band between plasma and red cells. Its thickness depends on leukocyte concentration, donor variability, and blood volume processed. In some samples, the buffy coat may be very thin, requiring careful visual inspection.
Good lighting and holding the tube against a contrasting background can help distinguish the layer clearly. Avoid moving or tilting the tube excessively once centrifugation is complete.
Step 5: Collect the Buffy Coat
- Use a pipette to carefully aspirate the buffy coat layer
- Minimize uptake of red blood cells
- Transfer to a fresh tube
Collection should be slow and controlled. It is better to collect slightly less material than to contaminate the buffy coat with red blood cells. If some plasma is collected along with the buffy coat, it can be removed during washing steps.
Using narrow pipette tips improves precision when aspirating thin layers.
Step 6: Optional Washing
- Add wash buffer
- Centrifuge at low speed (e.g., 300 × g for 10 minutes)
- Remove supernatant and resuspend
Washing helps remove residual plasma proteins and platelets, improving purity for downstream applications. After washing, resuspend the cell pellet gently to maintain viability.
At this stage, the buffy coat is ready for downstream cell isolation or enrichment workflows, including leukocyte separation, PBMC isolation, or targeted cell enrichment protocols.
Common Mistakes and How to Avoid Them
ven though buffy coat preparation is a straightforward procedure, small oversights can significantly affect yield, purity, and reproducibility. Most problems arise not from the protocol itself, but from how individual steps are handled. Understanding these common mistakes helps laboratories achieve more consistent and reliable results.
Using the Centrifuge Brake
One of the most frequent errors is applying the centrifuge brake at the end of the spin. Sudden deceleration creates turbulence inside the tube, disturbing the delicate separation between plasma, buffy coat, and red blood cells. This mixing makes the buffy coat harder to identify and collect cleanly. Always turn the brake off and allow the centrifuge to decelerate naturally to preserve clear layer boundaries.
Incorrect Centrifugation Speed
Centrifugation speed plays a critical role in forming a clean buffy coat. Speeds that are too high compress the layers tightly together, increasing red blood cell contamination. Speeds that are too low may fail to separate cells effectively, resulting in a diffuse or poorly defined buffy coat. Using validated settings—such as 200 × g for initial separation—ensures consistent and repeatable outcomes.
Over-Aspirating During Collection
Collecting the buffy coat requires patience and precision. Aspirating too aggressively or pulling material from too deep into the red blood cell layer reduces purity and introduces unwanted cells. Using a slow, controlled pipetting motion and collecting only the visible interphase helps maintain enrichment while minimizing contamination.
Delayed Processing After Centrifugation
Once centrifugation is complete, the separated layers are stable only for a limited time. Allowing tubes to stand too long can lead to diffusion between layers, making the buffy coat less distinct. Prompt collection immediately after centrifugation helps preserve separation quality.
Poor Blood Quality
Clotted, improperly anticoagulated, or aged blood often produces uneven or inconsistent layers. Platelet activation and cell aggregation increase over time, complicating separation. Whenever possible, work with fresh, well-mixed samples and ensure proper anticoagulation from the start.
By avoiding these common mistakes, laboratories can improve buffy coat purity, increase cell recovery, and achieve more reliable downstream results.
Using Buffy Coat for Downstream Cell Isolation and Enrichment
Once prepared, buffy coat serves as an excellent input for a wide range of cell isolation workflows.
Advantages for Cell Separation
- High leukocyte density reduces processing time
- Lower red blood cell burden simplifies enrichment
- Smaller volume improves handling efficiency
Suitable Applications
- Leukocyte isolation
- PBMC separation
- Rare cell enrichment
- Sequential isolation of multiple cell types
Because buffy coat contains concentrated immune cells, it is especially useful when isolating rare populations or performing multiple enrichment steps from the same sample. The remaining material can often be reused for additional isolations, maximizing sample value.
Conclusion
Preparing buffy coat from whole blood in your own lab is a practical, efficient approach that strengthens the foundation of many cell isolation workflows. With only standard centrifugation equipment and careful handling, laboratories can reliably generate leukocyte-enriched fractions without relying on external processing or complex preparation steps. This control over sample preparation allows researchers to work faster while maintaining consistency across experiments.
A clear understanding of how buffy coat forms, and how small details such as dilution, centrifugation speed, and gentle collection affect the outcome, helps reduce variability and cell loss. When performed correctly, buffy coat preparation delivers a concentrated source of immune cells that is easier to process than whole blood and better suited for downstream separation, enrichment, and functional analysis.
Compared to starting directly from whole blood, buffy coat preparation simplifies workflows, reduces processing time, and improves access to target cell populations. This is especially valuable when working with limited samples, rare cell types, or time-sensitive experiments where yield and viability matter most.
For laboratories focused on immune research, translational studies, or routine cell isolation, in-house buffy coat preparation is more than a convenience, it is a reliable, scalable starting point that improves efficiency, data quality, and confidence in downstream results.
 English
English French
French
 German
German
 Spanish
Spanish
 Belgium
Belgium
 Italian
Italian Brazil
Brazil Chinese Mandarin
Chinese Mandarin